Research
Peptide COA Explained: What Every Researcher Must Know
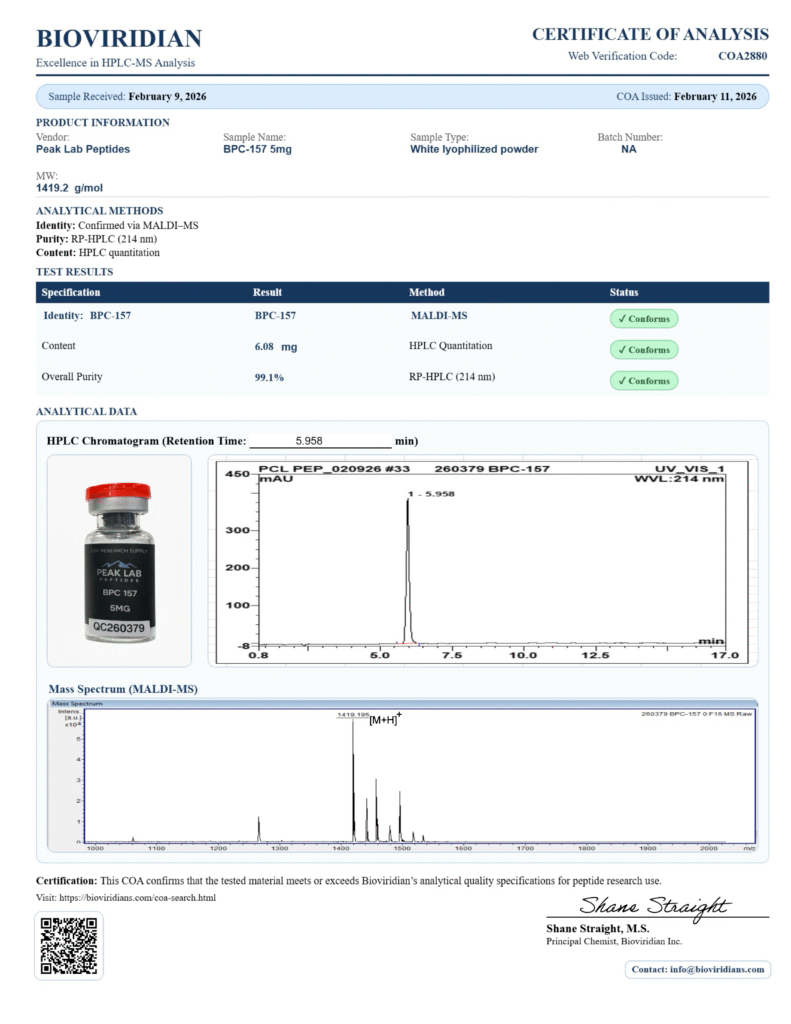
In laboratory research, a peptide COA (Certificate of Analysis) is one of the most important documents a researcher can review before working with a compound. Whether researchers are studying chemical compounds, biological materials, or experimental reagents, confidence in the underlying materials is foundational. One of the primary tools used to establish that confidence is a Certificate of Analysis, commonly referred to as a COA.
This article explains what a COA is, what it can and cannot tell you, and why it plays an important role in research settings.
What Is a Peptide COA?
A peptide COA (Certificate of Analysis) is a document issued by a laboratory that reports the results of analytical testing performed on a specific batch of material. It is typically generated after testing is completed and is tied to a unique lot or batch identifier.
In research contexts, a COA serves as a snapshot of measured properties at the time of testing. It provides transparency into how a material was analyzed and what parameters were evaluated, allowing researchers to better understand the characteristics of the material they are working with.
COAs are commonly used in academic, industrial, and preclinical research environments as part of standard documentation and quality control practices.
What COAs Measure
The exact contents of a COA vary depending on the material and the testing methods used, but most COAs focus on a few core categories:
1. Identity
Identity testing confirms that the material analyzed matches the expected chemical or molecular structure. For synthetic research peptides, two methods are most commonly used:
High-Performance Liquid Chromatography (HPLC) separates the components of a sample and produces a chromatogram showing the relative proportion of each detected substance. The main peak area relative to total peak area gives the purity percentage. HPLC is the standard method for peptide purity verification in research settings (PubMed: HPLC Analysis and Purification of Peptides).
Mass Spectrometry (MS) confirms the molecular weight and fragmentation pattern of the peptide, verifying that the correct sequence was synthesized. HPLC and MS are frequently used together — HPLC measures purity, MS confirms identity.
2. Purity
Purity measurements estimate the proportion of the target compound relative to other detectable substances in the sample. Purity is often reported as a percentage and is dependent on the sensitivity and scope of the analytical method used.
Different testing methods can yield different purity values, which is why understanding the methodology listed on a COA is important.
3. Contaminants or Impurities
Some COAs include analysis for specific impurities or contaminants, such as residual solvents, byproducts, or other detectable substances. The presence or absence of certain contaminants may be relevant depending on the nature of the research.
It is important to note that contaminant testing is typically limited to what was specifically tested for, not an exhaustive scan of all possible substances.
What COAs Do Not Measure
While COAs are useful, they are often misunderstood or overinterpreted. A COA has clear limitations.
A COA does not:
- Guarantee performance in any experimental system
- Predict biological, chemical, or experimental outcomes
- Assess suitability for a particular application beyond the tests listed
- Replace independent verification or replication
- Provide conclusions beyond the specific parameters tested
In other words, a COA reflects measured data, not outcomes, effects, or suitability for any particular use.
Common Misconceptions About Lab Testing and COAs
“A COA proves the material will behave a certain way.”
It does not. A COA reports analytical results, not experimental performance.
“High purity means fewer variables in research.”
Purity is one factor among many. Experimental conditions, handling, storage, and methodology all influence outcomes.
“A COA covers everything about a material.”
It only covers what was tested, using the methods listed, at a specific point in time.
“All COAs are the same.”
They are not. Different laboratories, methods, detection limits, and reporting standards can lead to meaningful differences between COAs.
Understanding these limitations helps prevent misinterpretation of analytical data.
Why COAs Matter in Research Settings
Despite their limitations, COAs remain an important part of responsible research practice.
They help researchers:
- Establish baseline documentation for materials used in studies
- Compare batches or lots over time
- Understand analytical methods applied to a material
- Support reproducibility and transparency in experimental design
- Maintain clear records for internal review or collaboration
In short, COAs contribute to traceability and accountability, both of which are essential in scientific work.
For readers interested in how COAs are presented and interpreted in practice, you can review additional information on our Certificate of Analysis page.
A Final Note on Interpretation
A Certificate of Analysis is a tool — not a conclusion. It provides structured data, but responsible interpretation requires context, methodological understanding, and an appreciation of experimental limitations.
Approaching COAs with a clear understanding of what they represent — and what they do not — is an important part of conducting and evaluating laboratory research responsibly.
This article is provided for informational purposes related to laboratory research and does not address or recommend use in humans or animals.
