Certificate of Analysis
Peak Lab Peptides — Certificate of Analysis (COA) Policy
Our Commitment to Quality & Transparency
At Peak Lab Peptides, we are committed to providing research-grade materials supported by clear quality-control practices. Our goal is to offer reliable documentation wherever possible, maintain consistent production standards, and continually expand access to batch-level testing data.
All materials are intended strictly for laboratory research use only and are not for human consumption.
Third-Party Testing Practices
Peak Lab Peptides utilizes third-party laboratories to support identity and purity verification on select batches. These tests may include, where applicable:
- HPLC analysis
- Mass spectrometry
- Microbial screening
- Structural verification
Testing frequency may vary based on production timelines, supplier scheduling, and batch volume.
Latests COAs
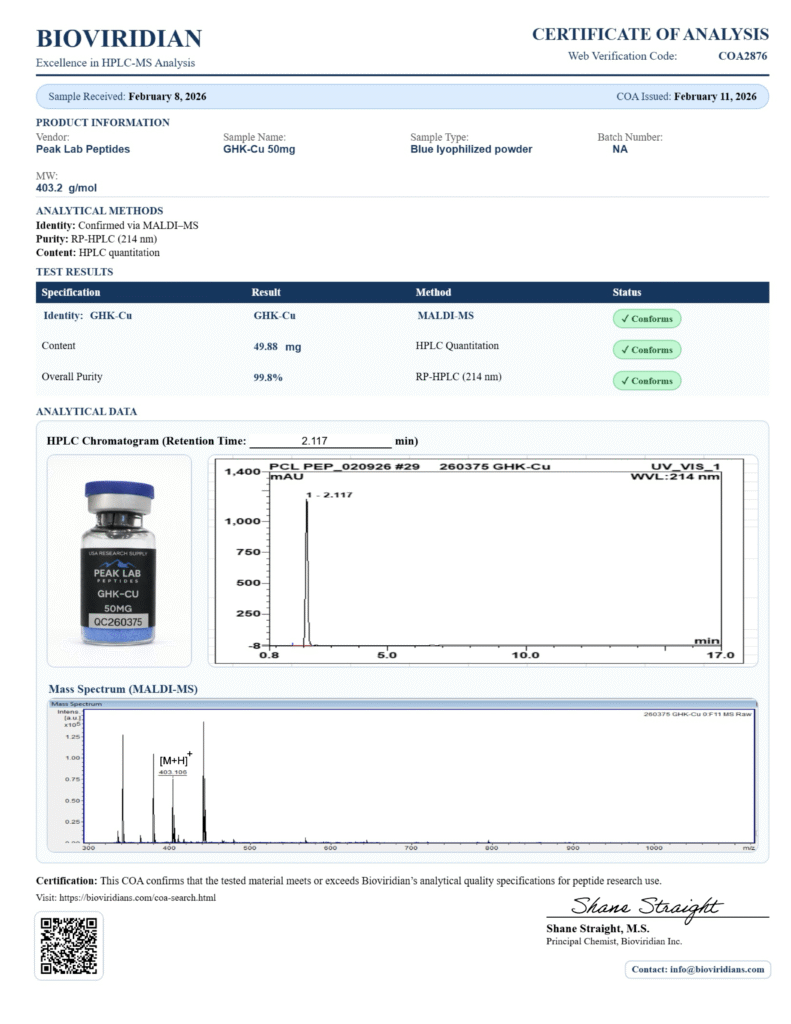
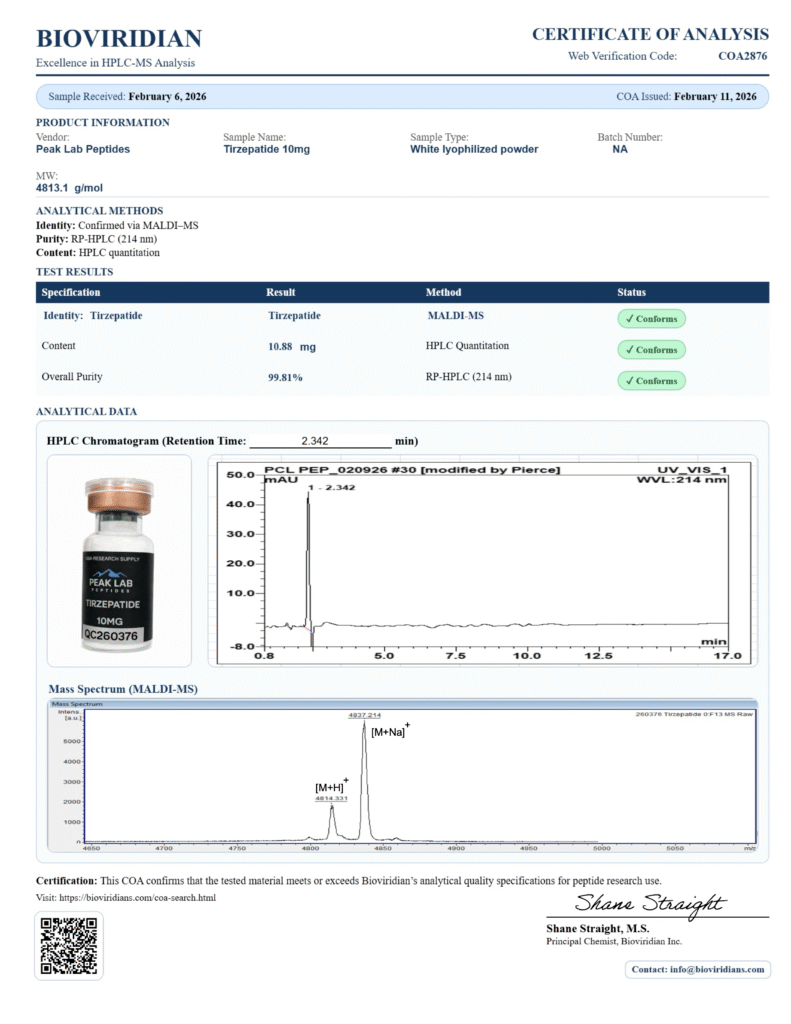
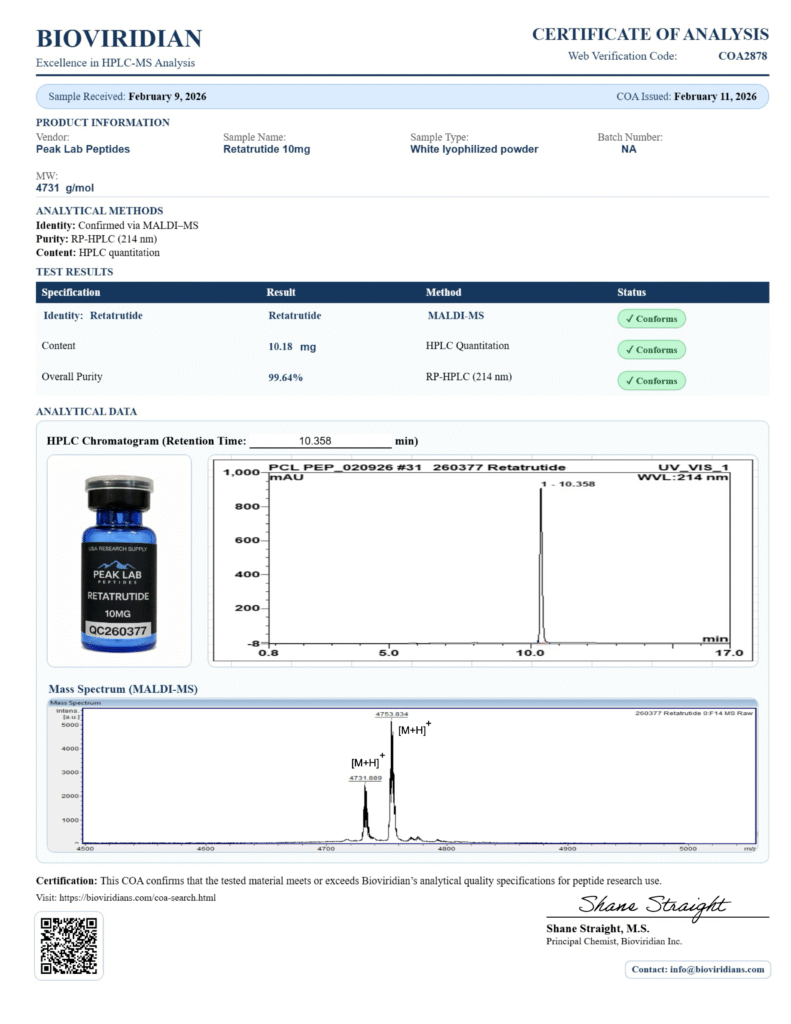
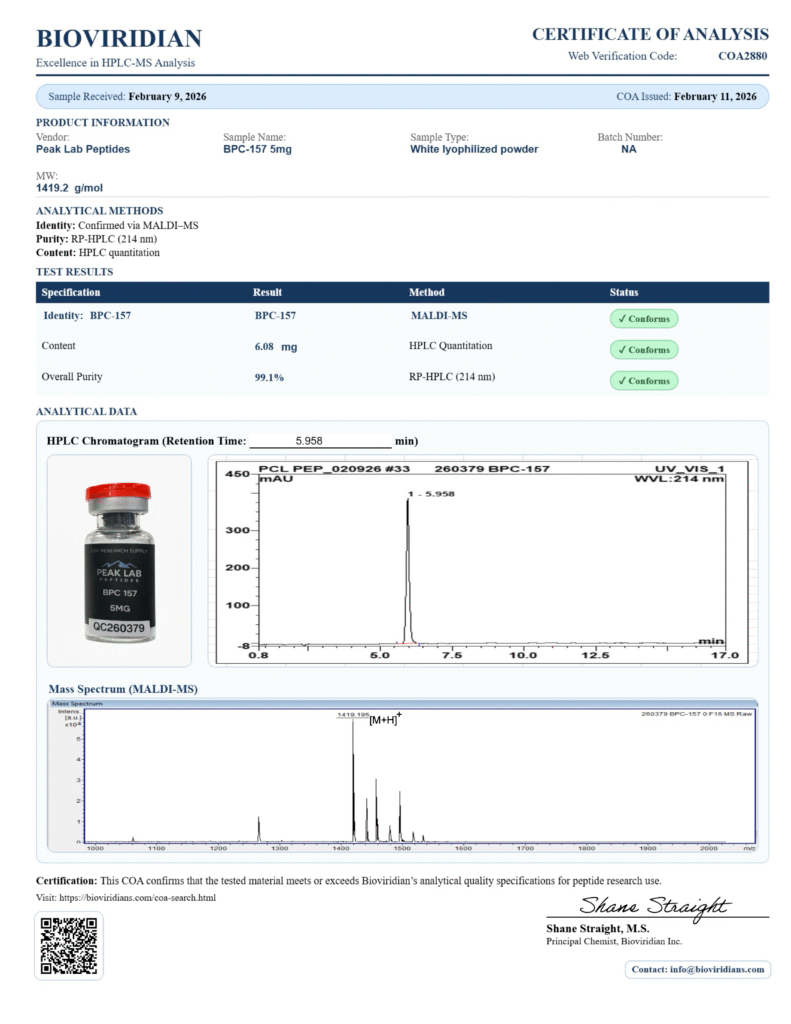
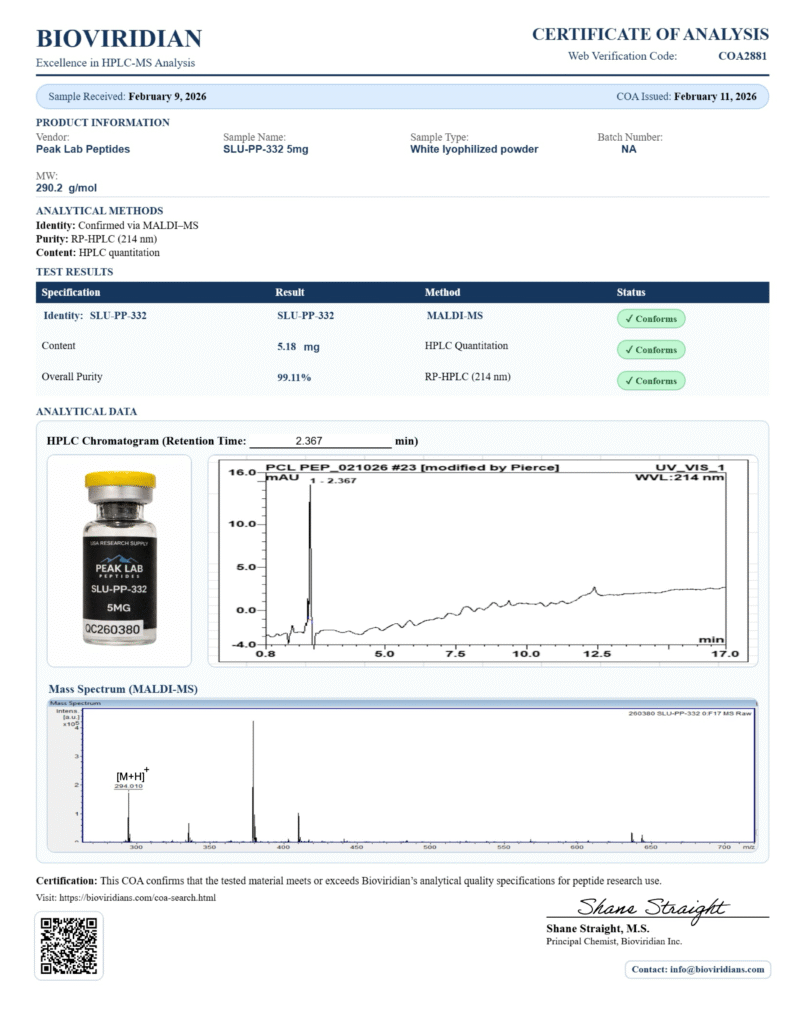
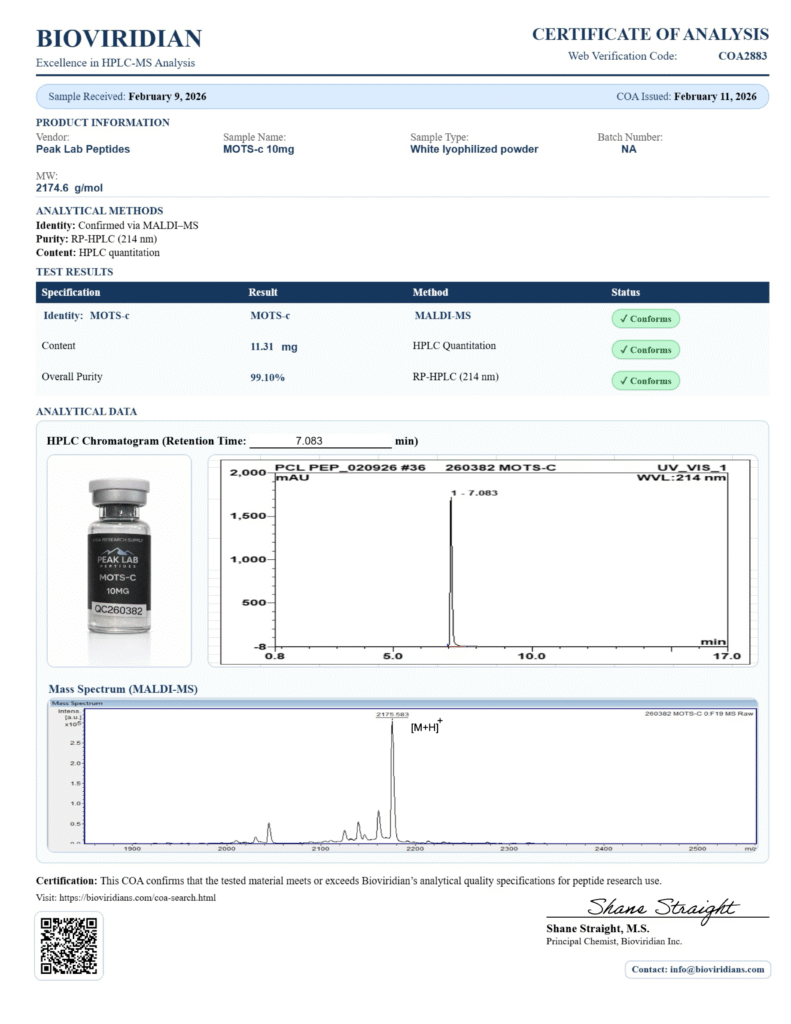
Availability of COAs
COA availability may differ by product and batch. Some products may have complete documentation available, while others may be in queue for testing or awaiting laboratory processing.
Current Policy:
COAs are available upon request for applicable batches.
Documentation may vary based on test availability, production cycles, and laboratory reporting times.
If a COA is not available for the specific batch shipped, Peak Lab Peptides can provide available documentation for the most recent comparable batch, when applicable.
How to Request a COA
To request a Certificate of Analysis for a product you have purchased:
- Email our support team at support@PeakLabPeptides.com
- Include your order number, product name, and lot/batch number (if visible on the vial)
- Our team will reply with the latest available documentation for that product or batch, if applicable
Typical response time: 1–2 business days
Ongoing Expansion of COA Coverage
Peak Lab Peptides is actively working to increase testing frequency and expand COA availability across the full catalog. As production capacity grows, additional batches will be submitted for third-party analysis to better support transparency and research reliability.
We will update this policy as additional COA coverage becomes available.
Quality-Control Statement
All products undergo internal quality-control checks prior to distribution. These assessments help ensure products meet labeling and identification standards, even in cases where third-party testing is still pending.
Peak Lab Peptides does not claim clinical validation, safety validation, or suitability for human use.
Regulatory Notice
All materials sold by Peak Lab Peptides are:
- For laboratory research use only
- Not intended for human or veterinary use
- Not for medical, therapeutic, or diagnostic use
By purchasing, customers acknowledge these conditions and agree to handle materials in accordance with applicable research and laboratory safety standards.
Contact Us
For COA requests, documentation inquiries, or quality-control questions, please contact:
Your trust matters — and we are committed to supporting transparent, reliable research.
