LL-37 (5mg)

$105.00 Original price was: $105.00.$84.00Current price is: $84.00.
In stock
Become a Member and save 20% on Every Order.
Description
LL-37 – 5mg (Lyophilized Peptide in 3ml Vial) – For Research Use Only
LL-37 is a human antimicrobial peptide derived from the cathelicidin family, extensively studied for its roles in innate immunity, antimicrobial defense, wound healing, and inflammation regulation. LL-37 exhibits broad-spectrum activity against bacteria, viruses, and fungi, and plays a key role in modulating the body’s immune and inflammatory responses.
In research, LL-37 is of particular interest in models exploring skin healing, immune signaling, tissue regeneration, and antimicrobial resistance.
Product Details:
Peptide: LL-37
Purity: >98%
Form: Lyophilized powder
Quantity: 5mg per vial
Vial Size: 3ml sterile glass vial
Storage: Store at -4°F (-20°C). After reconstitution, refrigerate at 36–46°F (2–8°C) and use within 30 days.
Grade: For research purposes only. Not for human consumption, therapeutic, or diagnostic use.
Potential Research Applications:
Antimicrobial and infection control models
Wound healing and skin regeneration studies
Immune system modulation and inflammation research
Studies on epithelial repair and barrier function
For laboratory research use only. This product is not intended to diagnose, treat, cure, or prevent any disease.
LL-37 is a human-derived antimicrobial peptide from the cathelicidin family that is widely studied for its role at the interface of host defense, inflammation, and tissue remodeling. In experimental models, LL-37 demonstrates broad-spectrum activity against bacteria, viruses, and fungi, while also interacting with immune and structural cells to influence inflammatory signaling, barrier integrity, and blood vessel formation.
As a research-only peptide, LL-37 is used to investigate innate immunity, inflammatory disease mechanisms, wound-repair biology, and tumor microenvironment dynamics. It is not approved for therapeutic, cosmetic, or diagnostic use in humans or animals.
LL-37 is the only known human cathelicidin-derived peptide and is generated by proteolytic processing of the CAP18 precursor protein. It is expressed in various epithelial surfaces and immune cells, especially at barrier sites such as skin, airways, and the gastrointestinal tract.
Key characteristics of LL-37 in research contexts include:
- Innate immune effector: Interacts with microbial membranes and host pattern-recognition systems.
- Immunomodulator: Can either enhance or restrain inflammatory responses depending on cell type, activation state, and local cytokine milieu.
- Tissue homeostasis mediator: Has been studied in relation to epithelial repair, angiogenesis, and extracellular matrix remodeling.
Because LL-37 shows context-dependent behavior, it serves as a versatile probe for exploring how a single peptide can coordinate antimicrobial defense with controlled inflammation and tissue repair.
7 reviews for LL-37 (5mg)
LL-37 and Inflammatory Diseases
LL-37 is frequently detected in inflamed tissues and has been evaluated in models of psoriasis, lupus, rheumatoid arthritis, atherosclerosis, and other immune-mediated conditions. Depending on the local environment, LL-37 has been observed to:
- Decrease keratinocyte apoptosis in certain skin models
- Increase interferon-alpha (IFN-α) production under specific stimulation conditions
- Influence chemotaxis of neutrophils and eosinophils
- Down-regulate toll-like receptor 4 (TLR4)–mediated signaling in selected systems
- Increase interleukin-18 (IL-18) production in some contexts
- Associate with reduced atherosclerotic plaque burden in certain experimental models
A notable feature is that LL-37 does not behave uniformly. For example, in T-cell research systems, LL-37 may enhance inflammatory actions when cells are not yet activated but may dampen inflammation once cells are already activated. This has led to the working hypothesis that LL-37 functions as a homeostatic modulator, helping fine-tune immune reactivity rather than driving a fixed pro- or anti-inflammatory outcome.
Antimicrobial Activity of LL-37
As part of the innate immune system, LL-37 is among the early-responding peptides during microbial challenge:
- Skin models: Baseline skin often shows low LL-37 levels, which can rise markedly upon exposure to microbial agents.
- Synergy with other peptides: LL-37 has been observed to act cooperatively with other endogenous peptides, such as human beta-defensin 2, in experimental infection models.
- LPS binding: LL-37 binds bacterial lipopolysaccharide (LPS), an essential component of gram-negative bacterial outer membranes, thereby disrupting microbial membrane integrity and contributing to direct antimicrobial effects.
- Gram-positive organisms: Despite its well-described interactions with LPS, LL-37 also demonstrates activity against gram-positive organisms and has been shown in vitro to enhance the effects of lysozyme against pathogens such as Staphylococcus aureus.
These properties make LL-37 a useful tool for studying multi-layered innate defense mechanisms in vitro and in vivo.
LL-37 and Lung Biology
In respiratory models, LL-37 has been studied in the context of airborne LPS exposure, toxic dust environments, and chronic airway disease:
- Epithelial response: LL-37 expression can increase in airway epithelial cells following exposure to irritants or microbial products.
- Wound closure and repair: Research indicates that LL-37 promotes airway epithelial cell migration and proliferation, supporting wound closure in in vitro and animal models.
- Angiogenesis and tissue homeostasis: LL-37 appears to contribute to coordinated repair responses by attracting structural cells and influencing local vasculature, underscoring its role as a potential homeostatic regulator in the airways.
These findings support the use of LL-37 as a model molecule for linking innate defense with epithelial repair in lung research.
LL-37 in Arthritis and Joint Inflammation
Elevated LL-37 levels have been reported in experimental models of joint inflammation, including those modeling rheumatoid arthritis:
- Presence in inflamed joints: LL-37 is detected in joint tissues and synovial fluid under inflammatory conditions, though current evidence does not support a direct pathogenic role.
- Protective hypotheses:
- LL-37–derived peptides have been shown in animal models to protect against collagen damage and reduce disease severity scores in inflammatory arthritis research systems.
- LL-37 has been associated with modulation of interleukin-32–driven inflammation, a cytokine linked to arthritis severity.
- Toll-like receptor pathways: LL-37 interacts with TLR pathways (e.g., TLR4), and ongoing work aims to clarify whether this interaction contributes to selective dampening of joint-destructive inflammatory responses.
Overall, LL-37 is being explored as a marker and potential modulator of inflammatory joint microenvironments rather than a primary cause of disease.
LL-37 and Intestinal Models
In gastrointestinal research, LL-37 has been evaluated for effects on epithelial barrier integrity, repair, and inflammation:
- Barrier maintenance: Cell culture studies show that LL-37 can enhance the migration of epithelial cells involved in maintaining the intestinal barrier.
- Apoptosis modulation: Under inflammatory conditions, LL-37 has been associated with reduced epithelial cell apoptosis, suggesting a possible protective effect on mucosal integrity.
- Combination with defensins: LL-37 appears to work alongside human beta-defensin 2 to promote wound closure and reduce TNF-related cell death in intestinal models.
These findings have led to interest in LL-37 as an experimental adjuvant in models of inflammatory bowel conditions, post-surgical healing, and antibiotic-associated gut injury—strictly at the research level.
LL-37, Cancer, and Blood Vessel Growth
The relationship between LL-37 and cancer is complex and context-dependent:
- Tumor microenvironment: In certain gastrointestinal and oral cancer models, LL-37 has been studied as part of vitamin D–responsive pathways in tumor-associated macrophages. These investigations look at how vitamin D–related signaling via LL-37 may influence local anti-tumor mechanisms.
- Angiogenesis: LL-37 can stimulate prostaglandin E2 (PGE2) production in endothelial cells, a mediator known to be involved in angiogenesis. This has implications for:
- Modeling pro-angiogenic responses in wound healing and ischemic tissue
- Exploring how altered angiogenesis may contribute to tumor growth or be targeted in cancer research
Through these lines of study, LL-37 is used as a tool to understand angiogenic control and the dual roles of inflammatory mediators in both repair and neoplastic processes.
Q1: What is LL-37 primarily used for in research?
A1: LL-37 is used to study innate immunity, antimicrobial defense, inflammatory signaling, tissue repair, and angiogenesis. It serves as a model peptide for understanding how one molecule can bridge host defense, immune modulation, and structural tissue responses.
Q2: Is LL-37 considered an antibiotic or a drug for treating infections?
A2: No. While LL-37 displays antimicrobial activity in experimental systems, LL-37 supplied for research is not approved or marketed as an antibiotic or therapeutic product. It is a research-only peptide used to explore host–pathogen interactions and innate immune mechanisms.
Q3: How does LL-37 affect inflammation in experimental models?
A3: LL-37 may either enhance or restrain inflammatory responses depending on cell type, activation state, and local cytokines. It has shown effects on cytokine production, cell recruitment, and pattern-recognition receptor signaling, making it a key peptide for investigating immune homeostasis rather than a simple pro- or anti-inflammatory factor.
Q4: Is LL-37 being investigated in connection with autoimmune or joint diseases?
A4: Yes. LL-37 has been detected in inflamed joints and has been studied in models of rheumatoid arthritis and other immune-mediated conditions. Current research focuses on its potential role as a modulator of inflammation and tissue damage, not as a confirmed therapeutic.
Q5: Can LL-37 be used for human therapy, cosmetic purposes, or self-administration?
A5: No. LL-37 distributed by research suppliers is intended strictly for laboratory use by qualified professionals. It is not approved for human or veterinary administration, cosmetic use, or any self-use application.
Storage & Handling
All peptides are supplied as sterile, lyophilized powder and are stable when handled correctly.
- On arrival: Store vials in a cool, dry place away from heat and direct sunlight.
- Long-term (powder): For optimal longevity, keep lyophilized peptides refrigerated to help maintain integrity.
- After reconstitution: Use an appropriate research diluent (for example, BAC water). Store the reconstituted solution in the refrigerator and use within 20–30 days for best stability.
Note: Minimize exposure to moisture and repeated freeze–thaw cycles. Follow your institution's safety procedures when handling research materials.
Peak Lab Peptides maintains quality-control processes and routinely performs third-party testing to support purity and identity verification. COAs are available upon request for applicable batches. Documentation may vary depending on production timelines.
We aim to make batch-level documentation available whenever possible. Our goal is to expand COA access across the full catalog as production capacity grows.
All products are for laboratory research use only and are not intended for human consumption.
Related Products
Acetic Acid 0.6%
Acetic Acid 0.6% – 10ml (Sterile Solution) – For Laboratory Use Only
Acetic Acid 0.6% is a sterile, aqueous solution commonly used in research and lab settings for pH adjustment, buffer preparation, and preservative applications. Its mild acidic properties make it suitable for studies involving topical antimicrobial effects, cellular environment modification, and reagent preparation. This solution is intended for controlled research environments requiring low-concentration acetic acid in a sterile, injectable-grade format.Product Details:
Compound: Acetic Acid 0.6% in sterile water Volume: 10ml multi-dose vial Form: Sterile solution Concentration: 0.6% w/v Storage: Store at 68–77°F (20–25°C); protect from light and contamination Grade: For laboratory research only. Not for human or animal use in therapeutic or diagnostic procedures.Potential Research Applications:
pH regulation and buffer system studies Topical antimicrobial and cytotoxicity research Reagent dilution and formulation preparation Laboratory protocol requiring acidic conditions For laboratory use only. This product is not intended to diagnose, treat, cure, or prevent any disease.AOD9604
AOD9604
AOD-9604 – 5mg (Lyophilized Peptide) – For Research Use Only
AOD-9604 is a modified fragment of human growth hormone (HGH), specifically amino acids 176–191, developed for its potential role in fat metabolism and lipolysis research. Unlike full-sequence HGH, AOD-9604 is studied for its ability to promote fat breakdown without influencing blood sugar or IGF-1 levels, making it a focus of metabolic and weight management studies.
Research has explored its effects on adipose tissue, body composition, and metabolic rate regulation.
Product Details:
-
Peptide: AOD-9604 (HGH Fragment 176–191)
-
Purity: >98%
-
Form: Lyophilized powder
-
Quantity: 5mg per vial
-
Storage: Store at -4°F (-20°C). After reconstitution, refrigerate at 36–46°F (2–8°C) and use within 30 days.
-
Grade: For research purposes only. Not for human consumption, therapeutic, or diagnostic use.
Potential Research Applications:
-
Fat metabolism and lipolysis studies
-
Obesity and weight management research models
-
Adipose tissue regulation investigations
-
Metabolic health and energy balance research
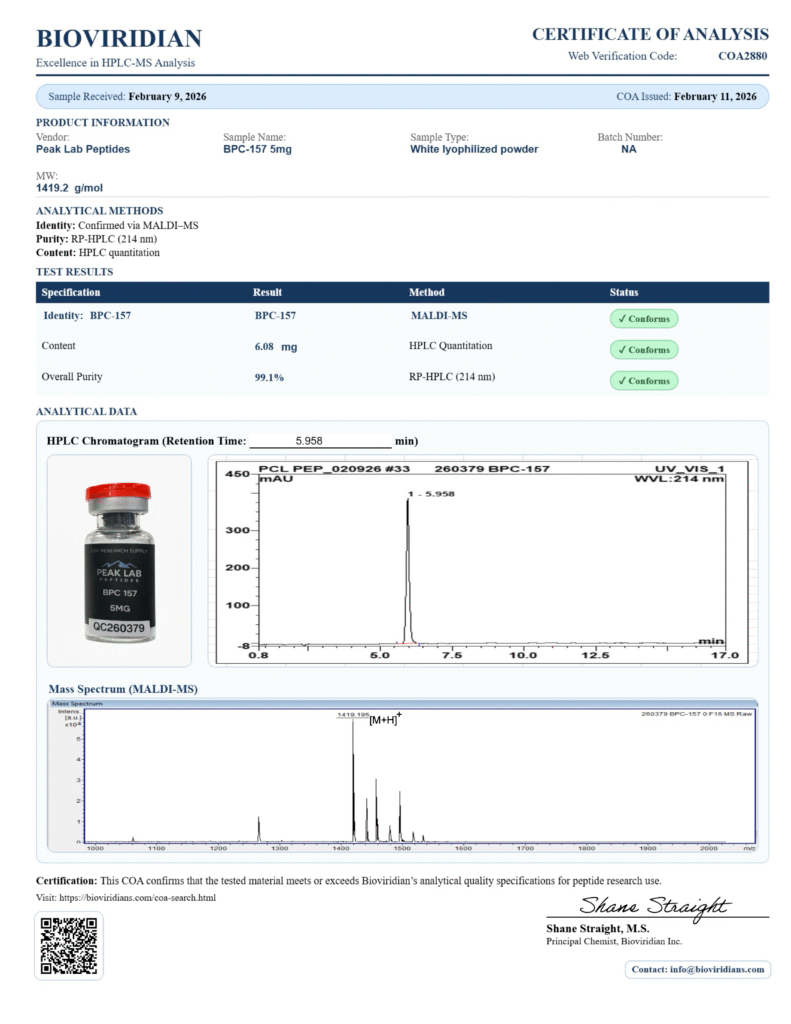
BPC-157, TB-500 – “Wolverine Stack”
BPC-157 and TB-500
BPC-157 + TB-500 – 10mg (Lyophilized Peptide Blend) – For Research Use Only
This advanced research peptide blend combines BPC-157 and TB-500 (Thymosin Beta-4 Fragment) — two well-studied peptides known for their potential in tissue repair, inflammation control, and accelerated recovery.
-
BPC-157 is a synthetic peptide derived from a gastric protein, studied for its possible role in gut health, tendon and ligament repair, and inflammation modulation.
-
TB-500 is a synthetic fragment of Thymosin Beta-4, researched for its potential to promote cell migration, angiogenesis, and muscle recovery.
Together, this combination is highly sought after in research models involving injury recovery, soft tissue repair, and anti-inflammatory studies.
Product Details:
-
Peptides: BPC-157 + TB-500 (5mg each unless otherwise specified)
-
Purity: >98%
-
Form: Lyophilized powder
-
Quantity: 10mg per vial
-
Storage: Store at -4°F (-20°C). After reconstitution, refrigerate at 36–46°F (2–8°C) and use within 30 days.
-
Grade: For research purposes only. Not for human consumption, therapeutic, or diagnostic use.
Potential Research Applications:
-
Injury recovery and soft tissue repair studies
-
Tendon, ligament, and muscle healing models
-
Anti-inflammatory and immune modulation research
-
Angiogenesis and cellular regeneration investigations
For laboratory research only. This product is not intended to diagnose, treat, cure, or prevent any disease.
CJC-1295 (WITH DAC)
CJC-1295 with DAC
CJC-1295 with DAC – 5mg (Lyophilized Peptide) – For Research Use Only
CJC-1295 with DAC (Drug Affinity Complex) is a long-acting analog of Growth Hormone-Releasing Hormone (GHRH), designed to promote sustained release of growth hormone (GH) in research models. The DAC modification significantly extends its half-life, allowing for prolonged activity and making it ideal for studies on muscle growth, recovery, anti-aging, and fat metabolism.
CJC-1295 with DAC is often paired in research with peptides like Ipamorelin for synergistic GH axis stimulation.
Product Details:
-
Peptide: CJC-1295 with DAC
-
Purity: >98%
-
Form: Lyophilized powder
-
Quantity: 5mg per vial
-
Storage: Store at -4°F (-20°C). After reconstitution, refrigerate at 36–46°F (2–8°C) and use within 30 days.
-
Grade: For research purposes only. Not for human consumption, therapeutic, or diagnostic use.
Potential Research Applications:
-
Sustained growth hormone release studies
-
Muscle growth, recovery, and anti-aging research models
-
Fat metabolism and body composition investigations
-
Long-acting GHRH analog research
For laboratory research only. This product is not intended to diagnose, treat, cure, or prevent any disease.
CJC-1295 No DAC
CJC-1295 No DAC
CJC-1295 No DAC – 5mg (Lyophilized Peptide) – For Research Use Only
CJC-1295 No DAC is a synthetic analog of Growth Hormone-Releasing Hormone (GHRH) designed for research on stimulating natural growth hormone (GH) release in a pulsatile manner. The “No DAC” (Drug Affinity Complex) version has a shorter half-life, allowing it to mimic the body’s natural GH secretion rhythm.
CJC-1295 No DAC is commonly studied in models focused on muscle growth, fat metabolism, anti-aging, and recovery when used alone or stacked with other GH secretagogues like Ipamorelin.
Product Details:
-
Peptide: CJC-1295 No DAC
-
Purity: >98%
-
Form: Lyophilized powder
-
Quantity: 5mg per vial
-
Storage: Store at -4°F (-20°C). After reconstitution, refrigerate at 36–46°F (2–8°C) and use within 30 days.
-
Grade: For research purposes only. Not for human use, medical, or diagnostic applications.
Potential Research Applications:
-
Growth hormone release studies
-
Muscle growth and recovery models
-
Anti-aging and longevity research
-
Fat metabolism and body composition investigations
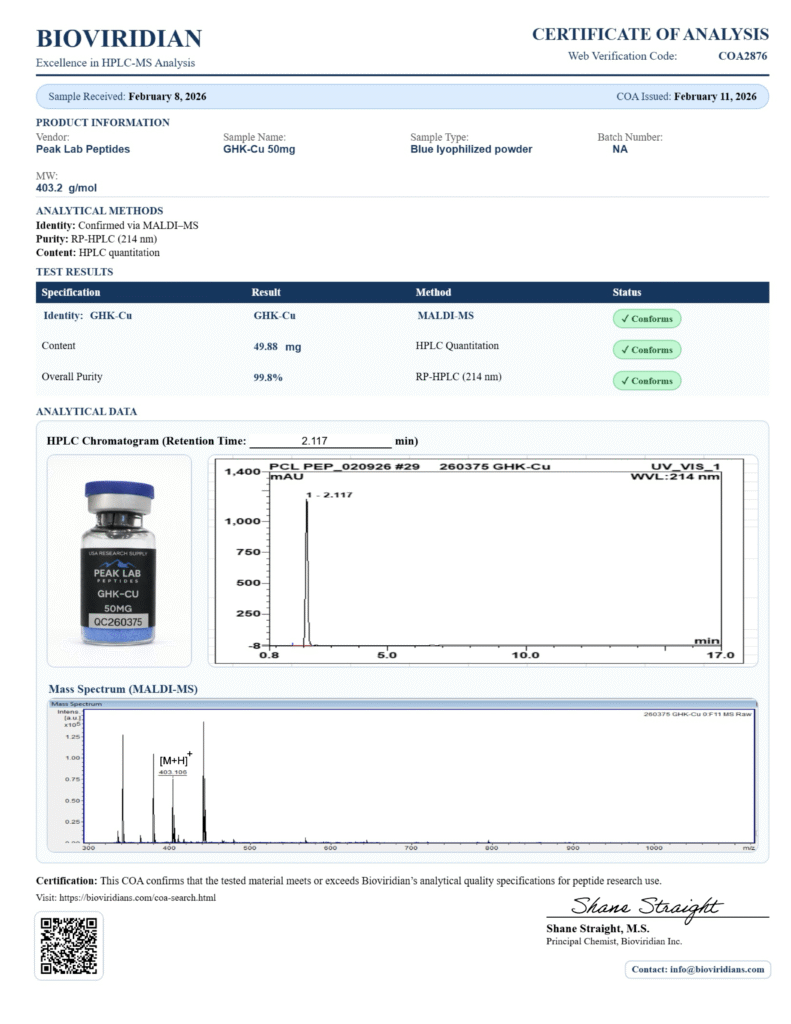
KLOW Blend 80mg – GHK-Cu, TB-500, BPC-157, KPV
KLOW – 80mg Peptide Blend (Lyophilized Powder) – For Research Use Only GHK-Cu • TB-500 • BPC-157 • KPV
KLOW is an advanced 4-in-1 research peptide blend combining 80mg of regenerative and anti-inflammatory compounds studied for their potential in tissue repair, immune modulation, and cellular recovery. Designed for comprehensive healing models, KLOW offers synergistic support across skin, muscle, and gut research applications. Active Ingredients (Per Vial): GHK-Cu – 50mg Studied for skin regeneration, wound healing, collagen stimulation, and hair follicle support. TB-500 – 10mg A fragment of Thymosin Beta-4, known for supporting angiogenesis, cell migration, and muscle recovery. BPC-157 – 10mg Derived from a gastric protein, BPC-157 is studied for gut repair, tendon/ligament healing, and anti-inflammatory effects. KPV – 10mg A tripeptide fragment with strong research interest for localized inflammation and immune modulation, particularly in the gut and skin. Product Details: Total Peptide Content: 80mg per vial: Lyophilized powder Purity: >98% Storage: Store at -4°F (-20°C). After reconstitution, refrigerate at 36–46°F (2–8°C) and use within 30 days. Grade: For research purposes only. Not for human consumption, therapeutic, or diagnostic use. Potential Research Applications: Soft tissue, muscle, and ligament repair Skin rejuvenation and wound healing models Inflammatory bowel and gut barrier integrity studies Anti-inflammatory and immune modulation research For laboratory research only. This product is not intended to diagnose, treat, cure, or prevent any disease.KPV
Product Details:
Peptide: KPV (Lys-Pro-Val) Purity: >98% Form: Lyophilized powder Quantity: 10mg per vial Storage: Store at -4°F (-20°C). After reconstitution, refrigerate at 36–46°F (2–8°C) and use within 30 days. Grade: For research purposes only. Not for human consumption, therapeutic, or diagnostic use. Potential Research Applications: Gut health and intestinal inflammation models (e.g., IBD, leaky gut) Skin inflammation and dermatitis research Cytokine modulation and immune response studies Localized anti-inflammatory peptide therapy research For laboratory research only. This product is not intended to diagnose, treat, cure, or prevent any disease.Melatonin
Melatonin
Melatonin – 100mg (Lyophilized Powder) – For Research Use Only
Melatonin is a naturally occurring hormone produced by the pineal gland, primarily known for its role in regulating the sleep-wake cycle (circadian rhythm). In research, Melatonin is studied for its potential effects on sleep regulation, antioxidant activity, immune modulation, and neuroprotection.
Studies have also explored its impact on oxidative stress and inflammation in various biological models.
Product Details:
-
Compound: Melatonin (N-Acetyl-5-methoxytryptamine)
-
Purity: >98%
-
Form: Lyophilized powder
-
Quantity: 100mg per vial
-
Storage: Store at -4°F (-20°C). After reconstitution, refrigerate at 36–46°F (2–8°C) and use within 30 days.
-
Grade: For research use only. Not for human consumption or therapeutic use.
Potential Research Applications:
-
Sleep and circadian rhythm studies
-
Antioxidant and oxidative stress research
-
Immune modulation investigations
-
Neuroprotection and anti-aging models
MT-2 (Melanotan 2) -10mg
Melanotan II (MT-2)
MT-2 (Melanotan 2) – 10mg (Lyophilized Peptide) – For Research Use Only
Melanotan 2 (MT-2) is a synthetic analog of the alpha-melanocyte-stimulating hormone (α-MSH), studied for its ability to activate melanocortin receptors involved in skin pigmentation, appetite regulation, and sexual function.
Research has focused on MT-2’s effects on melanin production, UV protection, libido, and energy balance in laboratory models.
Product Details:
-
Peptide: Melanotan 2 (MT-2)
-
Purity: >98%
-
Form: Lyophilized powder
-
Quantity: 10mg per vial
-
Storage: Store at -4°F (-20°C). After reconstitution, refrigerate at 36–46°F (2–8°C) and use within 30 days.
-
Grade: For research purposes only. Not for human consumption, therapeutic, or diagnostic use.
Potential Research Applications:
-
Melanogenesis and skin pigmentation studies
-
UV protection and tanning research models
-
Libido and sexual function investigations
-
Energy regulation and melanocortin receptor research
For laboratory research only. This product is not intended to diagnose, treat, cure, or prevent any disease.












Maria –
Highly satisfied with both product quality and service.
Dennis –
This store continues to meet expectations every time.
Jack –
This will remain my long-term choice for peptide purchases.
Kathleen –
Would confidently recommend this seller to others.
Tyler –
Everything about the ordering process feels smooth and professional.
Cynthia –
Delivery timelines have always been accurate and dependable.
Bob –
“Has anyone compared the effects of LL-37 with other immunomodulatory peptides like BPC-157 or KPV? I came across an interesting article discussing their potential roles in mast cell activation syndromes here: https://mindbodyneurology.com/bpc-157-and-kpv_peptides-for-mcas/. Specifically, I’m curious if LL-37 might share similar mechanisms in regulating inflammatory responses or if it operates through entirely different pathways. The product page mentions LL-37’s role in immune signaling and epithelial repair – does this overlap with how BPC-157 functions in gut healing? Would love to hear from researchers or anyone with firsthand lab experience working with these compounds.”